












We can help with...
BioChem QP Consulting Ltd. Registered Office 272 Bath Street, Glasgow G2 4JR
Tel +44 (0)771 897 1245 email enquiries@biochem-qp.com
IMP Importation for EU Clinical Trials
Your product’s gateway to Europe
Investigational Medicinal Products
Products for use in clinical trials are termed IMPs
(Investigational Medicinal Products) in the EU Good Manufacturing Practice
guidelines - the term also applies to placebos and comparator products, as well
as the active drug.
Getting IMPs manufactured in Third Countries into Europe
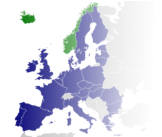
Any IMP batch that has been manufactured or QC tested in a so-called ‘third
country’ (i.e. a country outside the European Economic Area - coloured blue /
green above), must be certified by a Qualified Person before it can be used in a
European clinical trial. The QP has to be named on a manufacturer’s licence
within the EEA, with specific authorisation to import IMPs from outside Europe.
•
When you submit an application to conduct a clinical trial in Europe, you will
need to name an EEA-based importation site from where the imported
product will be released for onward distribution within the EEA
•
The QP at the importation site will need to provide a declaration of EU GMP
compliance for all 3rd country IMP manufacturing and testing sites, which
must be submitted along with the CTA (Clinical Trial Application) before you
can start your trial.
•
We will perform the necessary audits of third country manufacturing and QC
sites to provide the QP declaration for your CTA
•
We will import your IMP batches into Biostore UK’s MHRA-approved state of
the art controlled temperature storage facility in Scotland, with unrivalled
expertise in cold-chain storage (including liquid nitrogen and -70C). Once QP
certified by us, the IMP batch can move freely to any of your designated
distribution centres within Europe
•
We can also identify preferred partners for cold-chain shipping, IMP
re-labelling (including expiry updates), and clinical site distribution

Supply chain Risk
Assessment

Third Country site audits
against EU GMP

Liaison with clinical trial
sites

QP Declarations for IMPs
manufactured / tested in
Third Countries

Selection of couriers /
distrubution facilities


QP certification and batch
release of IMPs into Europe

IMPD authoring / reviews

Controlled temperature
storage, including ultracold


Specialists for sterile /
biological IMPs from USA









